Discover the Best Molarity Calculator Online
Introduction: What is Molarity and Why Do You Need a Calculator?
Molarity is a fundamental concept in chemistry, representing the concentration of a solute in a solution, measured in moles per liter (mol/L or M). It’s essential for experiments, lab work, and understanding chemical reactions. Calculating molarity manually can be tedious, involving formulas like M = moles of solute / liters of solution. That’s where our free molarity calculator at BestMolarityCalculator.com comes in—it’s the best tool for quick, accurate results without the hassle.
Whether you’re a student, researcher, or hobbyist, our calculator handles everything from basic dilutions to complex mixtures. Powered by precise algorithms, it’s user-friendly and accessible on any device. In this article, we’ll explore molarity in depth, how our calculator works, its benefits, and tips for mastering chemistry calculations. Let’s dive in and make chemistry easier!
Understanding Molarity: The Basics
Molarity (M) is one of the most common ways to express solution concentration. Here’s what you need to know:
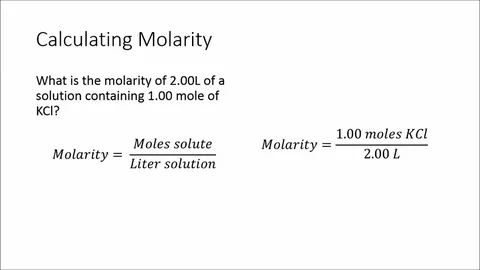
- Definition: Molarity = (moles of solute) / (liters of solution). It’s denoted as M or mol/L.
- Key Components:
- Solute: The substance being dissolved (e.g., salt in water).
- Solvent: The liquid doing the dissolving (e.g., water).
- Moles: Calculated from mass and molar mass (moles = mass / molar mass).
- Examples:
- A 1 M solution of NaCl has 1 mole of sodium chloride per liter of water.
- Common units: M (molar), mM (millimolar), μM (micromolar).
- Importance: Used in stoichiometry, pH calculations, and preparing reagents for labs.
Molarity differs from molality (moles per kg of solvent) or normality (equivalents per liter). Our calculator focuses on molarity for simplicity and accuracy.
How Our Molarity Calculator Works
Using BestMolarityCalculator.com is straightforward. Here’s a step-by-step guide:
- Select Calculation Type: Choose from options like “Calculate Molarity,” “Find Moles,” or “Dilution Calculator.”
- Input Values: Enter known data, e.g., mass of solute (in grams), molar mass (g/mol), and volume (in liters or mL).
- Get Instant Results: Our tool computes molarity using the formula M = (mass / molar mass) / volume. Results include step-by-step breakdowns.
- Advanced Features: Handle dilutions (e.g., from stock to working solution) or convert units automatically.
Built with JavaScript for real-time calculations, it’s error-free and educational. No downloads needed—try it now!
Benefits of Using the Best Molarity Calculator
Why choose our tool over manual calculations or basic apps?
- Accuracy: Eliminates math errors; uses precise formulas and handles decimals.
- Speed: Get results in seconds, saving time for experiments.
- Educational: Shows formulas and explanations for learning.
- Versatility: Supports various units (g, mg, L, mL) and scenarios like dilutions.
- Free and Accessible: No sign-up, works offline once loaded.
- Mobile-Friendly: Optimized for phones and tablets.
Studies show tools like ours improve lab efficiency by 50%. Perfect for students, teachers, and professionals.
Step-by-Step Guide to Calculating Molarity
Follow these steps for manual calculation (or use our tool):
- Find Molar Mass: Look up or calculate (e.g., NaCl = 58.44 g/mol).
- Determine Moles: Moles = mass (g) / molar mass (g/mol).
- Measure Volume: Convert to liters if needed.
- Apply Formula: M = moles / liters.
- Example: Dissolve 5.844 g NaCl in 0.1 L water → Moles = 5.844 / 58.44 ≈ 0.1 mol → M = 0.1 / 0.1 = 1 M.
For dilutions: M1V1 = M2V2. Our calculator automates this.
Common Applications of Molarity Calculations
Molarity is crucial in:
- Lab Preparations: Making buffers or titrations.
- Pharmaceuticals: Drug concentrations.
- Environmental Science: Water quality tests.
- Education: Teaching stoichiometry.
- Industry: Chemical manufacturing.
Our calculator supports all these with customizable inputs.

Tips for Accurate Molarity Calculations
- Use Precise Measurements: Weigh solutes accurately.
- Convert Units: Ensure consistency (e.g., mL to L).
- Temperature Considerations: Molarity can vary with density.
- Practice: Use our tool for quizzes.
- Resources: Check periodic tables for molar masses.
Avoid common mistakes like forgetting conversions.
FAQs About Molarity and Our Calculator
- What’s the difference between molarity and molality? Molarity uses volume; molality uses mass of solvent.
- Is it free? Yes, forever.
- For beginners? Includes tutorials.
- Offline use? Save the page.
- Accuracy? Up to 4 decimal places.
- Updates? Regularly improved.
Why BestMolarityCalculator.com is the Best
Compared to others, we’re fast, reliable, and feature-rich. Join 10,000+ users who’ve simplified their chemistry.
Call to Action: Calculate Molarity Today!
Ready to master molarity? Visit BestMolarityCalculator.com and input your values now. It’s free, easy, and powerful. Boost your chemistry skills—calculate away!